Genomics: Insight

Can modifying immune response genes prevent allo-HSCT patients from getting ARDS?
Acute Respiratory Distress Syndrome, otherwise known as ARDS, is a life-threatening syndrome that admits fluid into the lungs that builds up in your alveoli, tiny air sacs in your lungs which exchange oxygen and carbon dioxide. This prevents a sufficient amount of oxygen from entering your bloodstream that is necessary for your organs to function properly [2]. ARDS has significant long-term consequences, such as physical and cognitive impairment and a high mortality rate of 30-40% cause of death in adult intensive care units [3].
Introduction
A respiratory research paper, published by BMC, compares RNA expression patterns in oxygenated blood between ARDS patients who have received allogeneic hematopoietic stem cell transplantation (allo-HSCT), a treatment for blood cancer, and those who have not [1]. Identifying RNA expression patterns helps us understand which genes are upregulated or downregulated and whether certain genes are modulated. Allo-HSCT, is a procedure in which cancer patients with faulty immune systems are treated with specialized cells [4]. These specialized cells grow into blood cells and develop a new immune system after the previous immune system is destroyed by chemotherapy [5]. Patients with blood cancer undergoing allo-HSCT are an important subgroup with increased risk of ARDS and high mortality rates. The aim of this study is to identify differences in gene expression between ARDS subjects who received and didn’t receive allo-HSCT. By identifying these differences, we can target specific gene regions to prevent the development of ARDS in allo-HSCT patients.
...underscoring the need for a better understanding of these different populations.
Method
Researchers used blood samples of 11 ARDS patients to profile gene expression that were taken within 5 days of ICU admission, and the subjects were selected from similar age, sex, and race in order to reduce sample heterogeneity and produce reliable results. These 11 patients consisted of 5 people who had undergone allo-HSCT and 6 people who had not received the transplant. The study included 12 additional patients with HSCT sepsis and non-HSCT sepsis in order to identify differences between the ARDS groups that were not caused by HSCT status. Gene expression of ARDS patients and post allo-HSCT ARDS patients were compared using RNA sequencing. RNA sequencing is performed by isolating and purifying RNA, converting the RNA to cDNA, also known as complementary DNA, which is a processed copy of RNA, and analyzing the sequences resulting from the cDNA [6]. This displays the transcriptome of gene expression patterns encoded within RNA [7].
The overall study design is shown below:
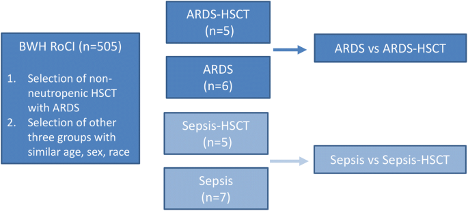
Subjects with sepsis were also included in order to identify differences between the ARDS groups that were not caused by HSCT status.
Results
Through RNA sequencing, 687 differentially expressed genes were identified between ARDS-HSCT and ARDS. 520 of these genes were upregulated and 167 were downregulated. After filtering out the genes also expressed in sepsis-HSCT and sepsis, 483 genes were found to be unique to ARDS-HSCT. Interestingly, some of the differentially expressed genes with the strongest differences in pathways, such as IFI44L, OAS3, LY6E, OAS2, and USP18, were related to immune responses to pathogens and type I interferon signaling, all of which were upregulated in ARDS. Though type I interferons increase cellular resistance to viral infection by initiating lung inflammatory responses, this inflammation sometimes contributes to the disease severity [8].
Modulation of interferon signaling pathways represents a treatment approach for HSCT subjects...
What these results suggest is that there is an immune dysregulation (malfunction of the immune system) that makes patients more susceptible to ARDS-HSCT. High and low blood expression of MX1, IFIT1, and ISG15, a set of interferon-stimulated genes, were found in patients with dire outcomes. In another study, analysis of the expression of monocyte genes, a type of white blood cell in peripheral blood, revealed increased interferon signaling in patients who had better outcomes compared to patients who had unfavorable outcomes [9]. This suggests that modulating interferon signaling pathways could potentially become a more targeted treatment method to HSCT subjects. This study and existing literature suggest that by modulating interferon signaling pathways, the immune response that triggers the development of ARDS after allo-HSCT could potentially be modified, preventing allo-HSCT patients from developing ARDS.
Conclusion
Previous studies identified gene-expression signatures in sepsis patients that were developing ARDS and discovered that ARDS patients had drastic increases in immune response pathways, a set of processes that triggers an immune response to a foreign substance [10], compared to healthy individuals. However, none of these studies characterized ARDS subgroups, which makes this study important because characterizing subgroups may unveil changes in gene expression that result in similar clinical symptoms.
This study identified differences in gene expression of ARDS and ARDS-HSCT, providing a potential treatment method of ARDS and proving the need for identification of ARDS subgroups in order to treat ARDS. The findings revealed changes in immune response and interferon signaling pathways, suggesting that dysregulated interferon signaling is the cause of lung injury after allo-HSCT. Both previous studies and this study support the possibility of modifying immune deficiency so that patients who undergo allo-HSCT are not at risk of lung injury and ARDS. Further research of treatment by modifying immune-deficiency serves as great hope for the future of ARDS.
References
- Englert, Joshua. “Whole Blood RNA Sequencing Reveals a Unique Transcriptomic Profile in Patients with ARDS Following Hematopoietic Stem Cell Transplantation.” Respiratory Research, BioMed Central, 1 Jan. 1970.
- “ARDS.” Mayo Clinic, Mayo Foundation for Medical Education and Research, 13 June 2020.
- Bellani, Giacomo. “Epidemiology, Patterns of Care, and Mortality for Patients With Acute Respiratory Distress Syndrome in Intensive Care Units in 50 Countries.” JAMA, U.S. National Library of Medicine, 2016.
- “What Is Hematopoietic Stem Cell Transplantation?” Medical News Today, MediLexicon International, 2017.
- Schley, David. “7 Things You Need to Know about HSCT.” Multiple Sclerosis Society UK, 2018.
- Cresko Lab. “Seqlopedia.”.
- MacKenzie, Ruairi. “RNA-Seq: Basics, Applications and Protocol.” Genomics Research from Technology Networks, 2018.
- Makris, Spyridon. “Type I Interferons as Regulators of Lung Inflammation.” Frontiers in Immunology, Frontiers Media S.A., 10 Mar. 2017, www.ncbi.nlm.nih.gov/pmc/articles/PMC5344902/.
- Morrell, Eric. “Peripheral and Alveolar Cell Transcriptional Programs Are Distinct in Acute Respiratory Distress Syndrome.” American Journal of Respiratory and Critical Care Medicine, 2017, www.atsjournals.org/doi/10.1164/rccm.201703-0614LE.
- The Jackson Laboratory. “Immune Response-Regulating Signaling Pathway.” Immune Response-Regulating Signaling Pathway Gene Ontology Term (GO:0002764), 2020.
About the Author
Beatrice Park is a 15-year-old student who attends Northside College Preparatory High School. She aspires to pursue research in the respiratory field and is fascinated by the role of genomics in respiratory diseases and syndromes.